
2018
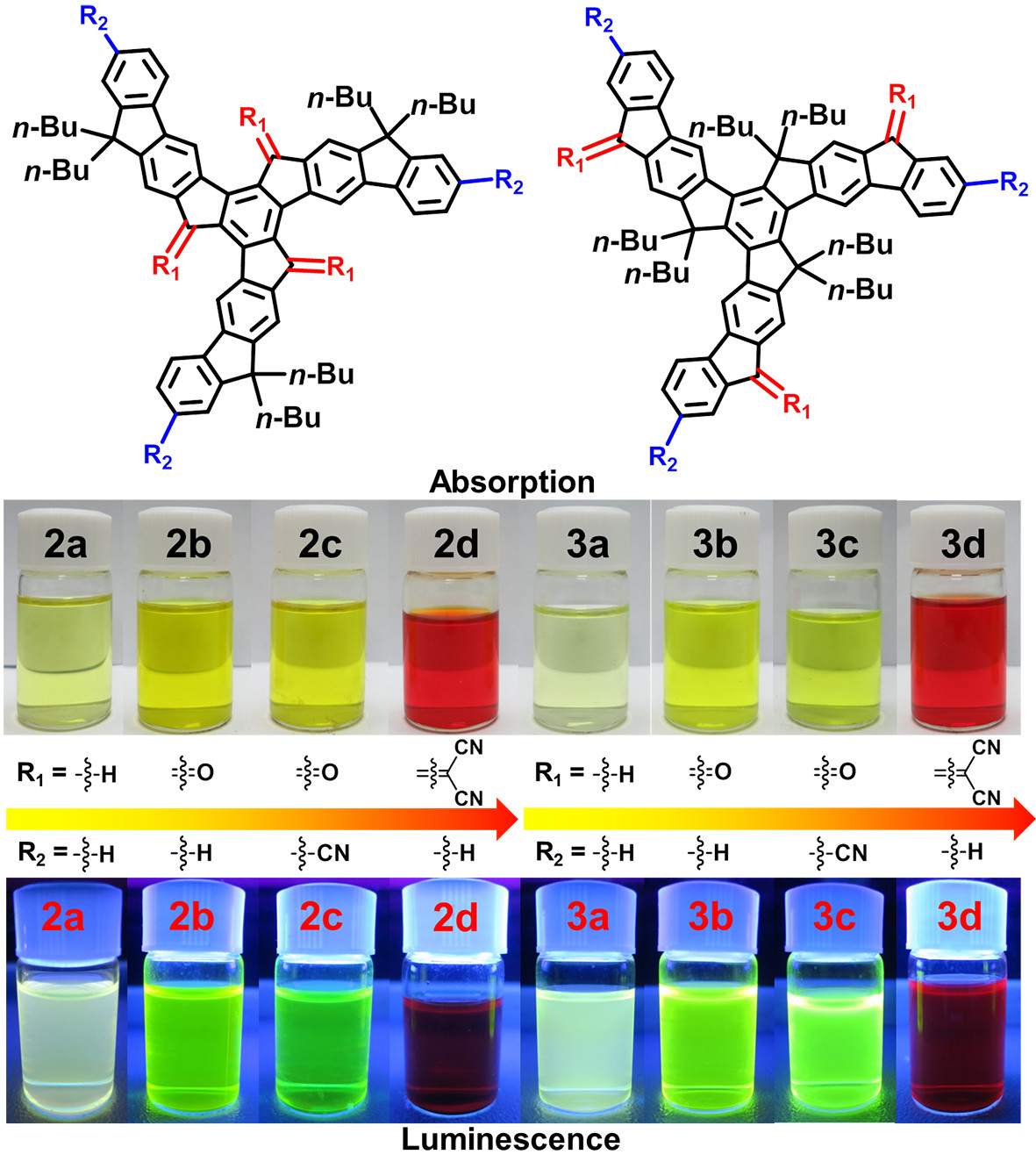
17 Oct 2018, Chem. Eur. J.
Jun-Bo Chen, Cen Zhou, Ru-Qiang Lu, Xin-Chang Wang, Hang Qu, Mithu Saha, Hao-Liang Liu, Hui Zhang and Xiao-Yu Cao*
Triindolo‐truxene, a C3‐symmetric molecule with a large π‐conjugated plane, has six methylene carbon atoms and three aromatic carbon atoms that can be facilely functionalized. Herein, butyl, carbonyl, cyano, and/or malononitrile groups were introduced at six methylene carbon atoms (6‐, 14‐, 22‐ or 8‐, 16‐, 24‐positions) and/or three aromatic carbon atoms (2‐, 10‐, and 18‐positions) of triindolo‐truxene to produce eight derivatives. Their photophysical properties, electrochemical properties, and molecular assembly can be effectively modulated by substituents and substitution patterns. Incorporation of electron‐deficient groups led to redshifts in both the absorption and emission of these derivatives and also lowered their HOMO and LUMO levels. Different substitution patterns resulted in the different intramolecular donor–acceptor interactions. Electron‐deficient substituents at the methylene carbon atoms in the 6‐, 14‐, and 22‐positions led to intramolecular charge transfer from the fluorene arms to the truxene core, whereas the corresponding substitutions at the methylene carbon atoms in the 8‐, 16‐, and 24‐positions resulted in intramolecular charge transfer from the truxene core to the fluorene arms. The molecular packing in single crystals and molecular aggregation in solution are also influenced by the substituents and substitution patterns. This work provides a straightforward strategy to alter the properties of triindolo‐truxene.
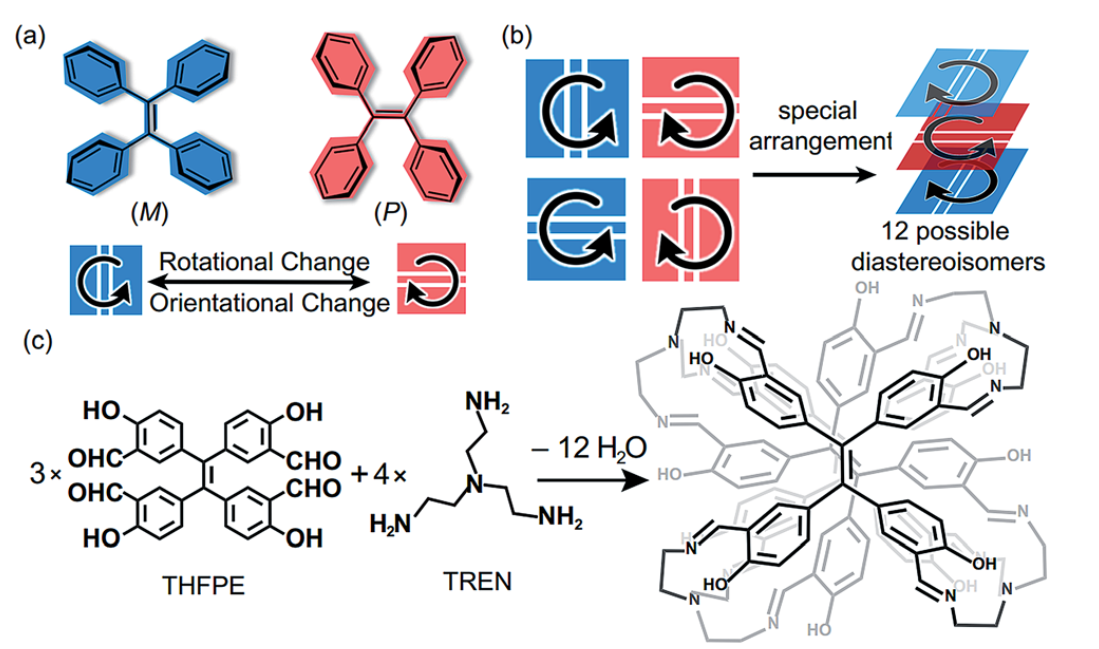
14 Sep 2018, Chemical Science
Hang Qu, Xiao Tang, Xinchang Wang, Zhihao Li, Zheyu Huang, Hui Zhang, Zhongqun Tian and Xiaoyu Cao *
Chiral tetraphenylethylene (TPE) derivatives have great potential in chiral recognition and circularly polarized luminescence. However, they were mainly constructed through introducing chiral substituents at the periphery of the TPE moiety, which required additional chemical modifications and limited the variety of chiralities of products. Herein, we constructed a series of chiral face-rotating sandwich structures (FRSs) through restricting the phenyl flipping of TPE without introducing any chiral substituents. In FRSs, the complex arrangements of TPE motifs resulted in a variety of chiralities. We also found that non-covalent repulsive interactions in vertices caused the facial hetero-directionality of FRSs, and the hydrogen bonds between imine bonds and hydroxy groups induced excited-state intramolecular proton transfer (ESIPT) emission of FRSs. In addition, the fluorescence intensity of FRSs decreases with the addition of trifluoroacetic acid. This study provides new insights into the rational design of chiral assemblies from aggregation-induced emission (AIE) active building blocks through restriction of intramolecular rotation (RIR).
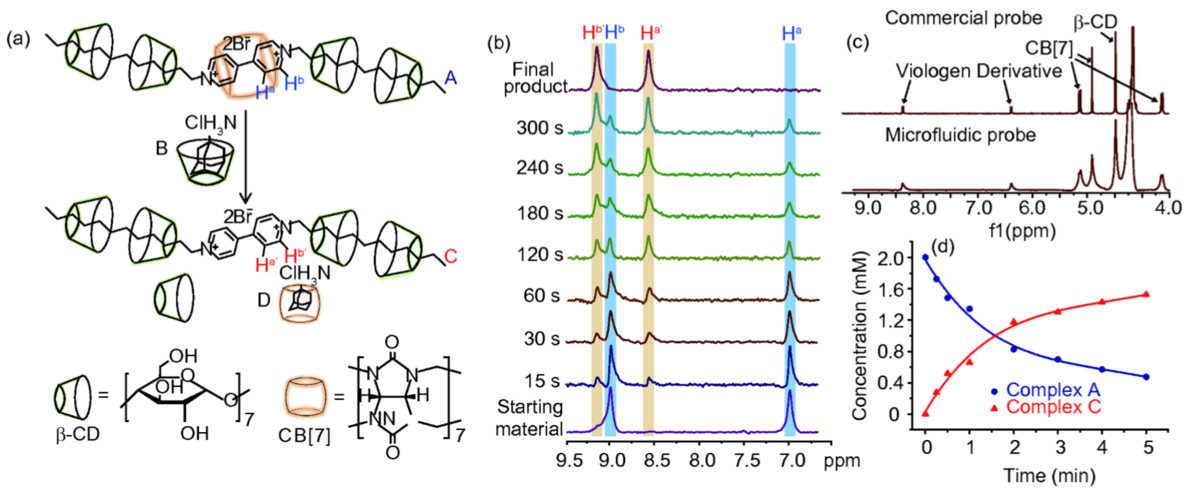
13 Sep 2018, Science China Chemistry
Hongxun Fang, Yibin Sun, Xinchang Wang, Manvendra Sharma, Zhong Chen, Xiaoyu Cao*, Marcel Utz* and Zhongqun Tian*
Microfluidic-NMR spectroscopy has been extended to study the kinetics in supramolecular chemistry and molecular assembly. Kinetics of a multicomponent host-guest supramolecular system containing viologen derivatives, β-cyclodextrins and cucurbit [7]urils are studied by a PMMA based microfluidic chip combined with a dedicated transmission line probe for NMR detection. By combining microfluidic technology with NMR spectroscopy, the amount of material required for a full kinetic study could be minimized. This is crucial in supramolecular chemistry, which often involves highly sophisticated and synthetically costly building blocks. The small size of the microfluidic structure is crucial in bringing the time scale for kinetic monitoring down to seconds. At the same time, the transmission line NMR probe provides sufficient sensitivity to work at low (2 mM) concentrations.
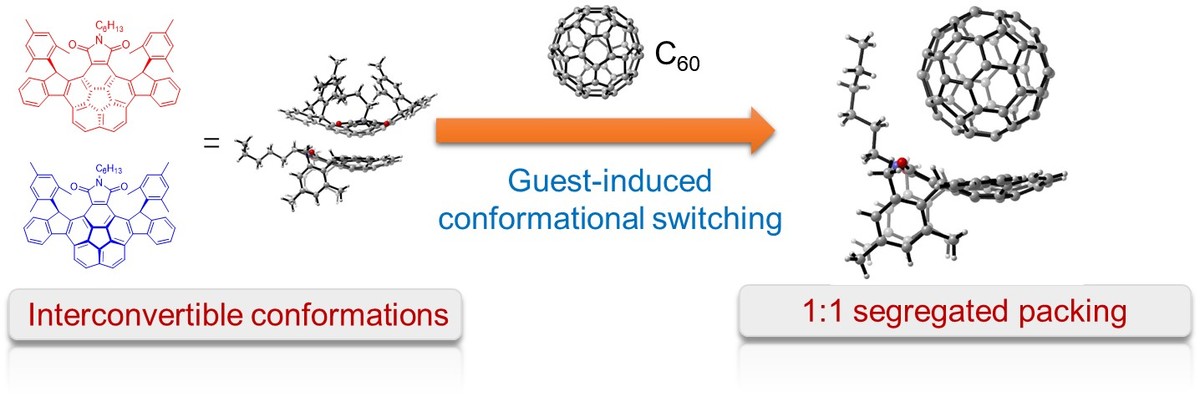
02 Aug 2018, Asian Chemical Editorial Society
Ru-Qiang Lu, Shuang Wu, Yue-Hua Bao, Lin-Lin Yang, Hang Qu, Mithu Saha, Xiao-Ye Wang, You-Zhen Zhuo, Binbin Xu, Jian Pei, Hui Zhang, Wengui Weng, and Xiao-Yu Cao*
A pair of interconvertible stereoisomers of imide‐fused corannulene derivatives was mixed with C60, which resulted in cocrystallization into a 1:1 segregated packing motif through concave–convex π–π interactions. Only one conformation was observed in the cocrystal owing to guest‐induced conformational switching. The 1D assemblies of the complex showed promising applications in organic electronics.
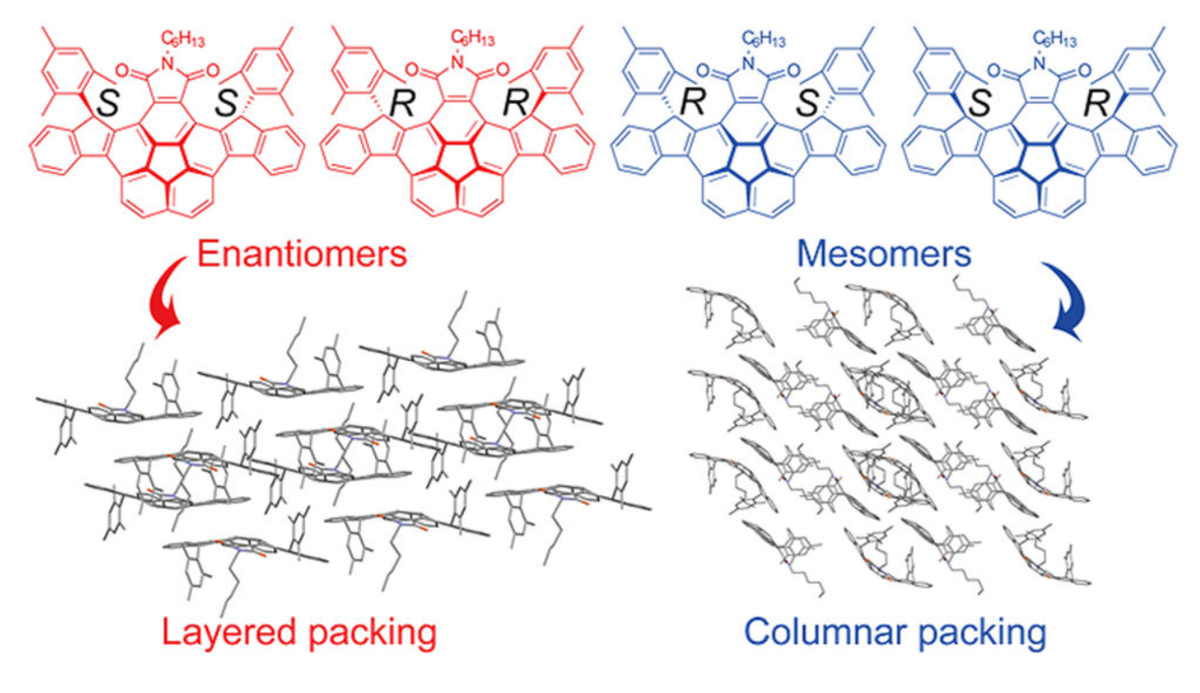
10 Jul 2018, Cryst. Growth Des
Ru-Qiang Lu, Yuxiu Liu, Shuang Wu, Mithu Saha, Hang Qu, Rui Chen, Lin-Lin Yang, Xiao-Ye Wang, Yuchen Wang, Wengui Weng, Yi Zhao,* and Xiaoyu Cao*
Two chiral tertiary carbon centers bearing one mesityl group at each center are introduced into the molecular backbone of imide-fused corannulene derivatives to produce four stereoisomers (i.e., (S, S), (R, R), (R, S), or (S, R) configurations on two chiral carbons) in one pot, which are separated into two portions through column chromatography over silica gel. Portion 1, containing a pair of enantiomers ((S, S) and (R, R)), adopts layered packing in the crystal. Portion 2, consisting of a pair of mesomers ((R, S) and (S, R)), exhibits columnar packing in their cocrystal. Theoretical calculations are performed on these two packing motifs, revealing that Portion 1 displays hole-dominated transport, whereas Portion 2 shows electron-dominated transport.
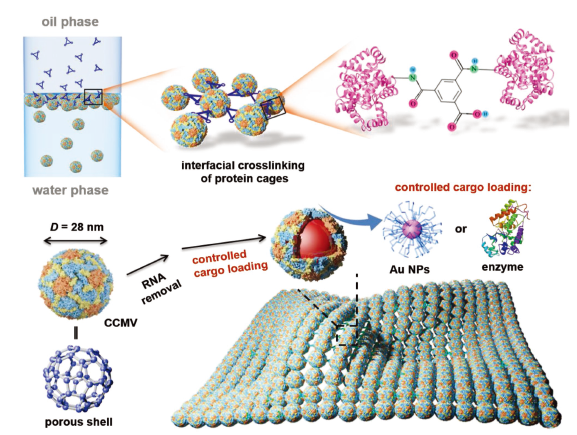
22 June 2018, Advanced Functional Materials
A. J. Liu, W. Zhu, M. V. de Ruiter, S. J. Maassen, R. M. Putri, L. L. Yang*, Jeroen J. L. M. Cornelissen*
Hybrid thin films with a high loading and homogeneous dispersion of functional nanoparticles (and/or molecules) find applications in (bio)‐sensors and electronic devices. The fabrication of such hybrid thin films, however, suffers from the complex and diverse surface and physicochemical properties of individual nanoparticles. To address this challenge, a facile and general strategy toward compartmentalized thin films through the interfacial cross‐linking of viral protein cages is reported. Employing these protein cages, gold nanoparticles, as well as enzyme horseradish peroxidase, are encapsulated into virus‐like particles and then cross‐linked into thin films with a thickness varying from monolayer to submicron dimensions. These compartmentalized thin films not only ensure that the cargo is homogeneously dispersed, but also display good catalytic activity. This strategy is, in principle, applicable for a wide range of (bio)‐organic nanocontainers, enabling the versatile fabrication of 2D thin films with extensive application prospects.
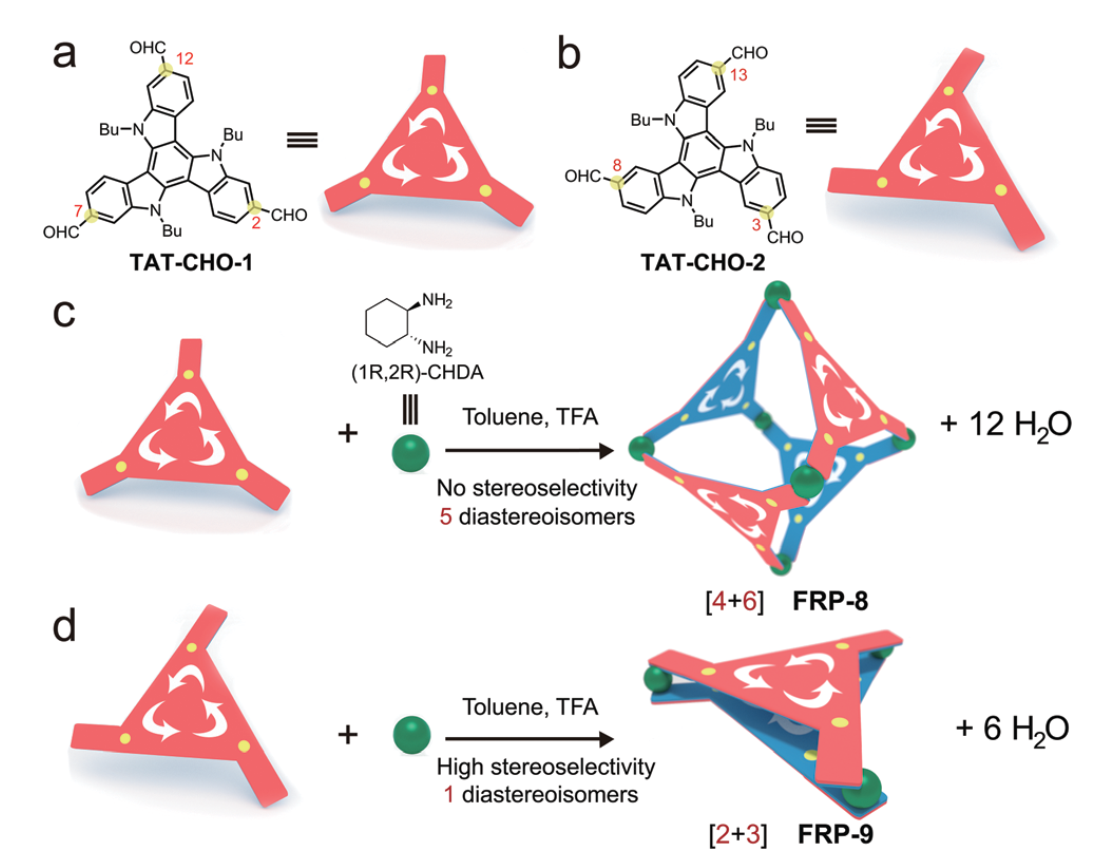
09 Apr 2018, Chemical Communications
Pei Zhang, Xinchang Wang, Wei Xuan, Pixian Peng, Zhihao Li, Ruqiang Lu, Shuang Wu, Zhongqun Tian and Xiaoyu Cao *
We constructed a series of novel chiral molecular face-rotating polyhedra (FRP) from two 10,15-dihydro-5H-diindolo[3,2-a:3′,2′-c]carbazole (triazatruxene) derivatives and trans-1,2-cyclohexane diamine, and investigated how facial interactions and the positions of substituents determine the diastereoselectivity and geometry of the final assemblies.
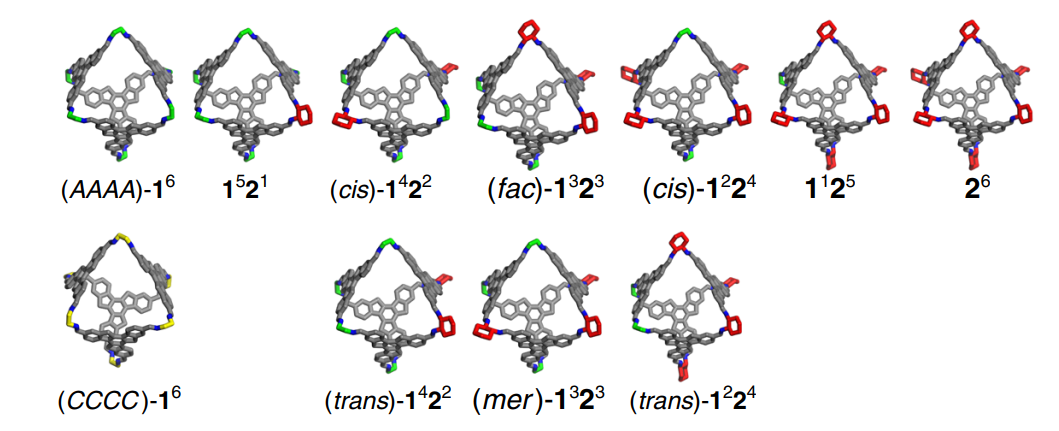
05 Feb 2018, Nature Communications
Yu Wang, Hongxun Fang, Ionut Tranca, Hang Qu, Xinchang Wang, Albert J. Markvoort, Zhongqun Tian and Xiaoyu Cao*
Chiral amplification in molecular self-assembly has profound impact on the recognition and separation of chiroptical materials, biomolecules, and pharmaceuticals. An understanding of how to control this phenomenon is nonetheless restricted by the structural complexity in multicomponent self-assembling systems. Here, we create chiral octahedra incorporating a combination of chiral and achiral vertices and show that their discrete nature makes these octahedra an ideal platform for in-depth investigation of chiral transfer. Through the construction of dynamic combinatorial libraries, the unique possibility to separate and characterise each individual assembly type, density functional theory calculations, and a theoretical equilibrium model, we elucidate that a single chiral unit suffices to control all other units in an octahedron and how this local amplification combined with the distribution of distinct assembly types culminates in the observed overall chiral amplification in the system. Our combined experimental and theoretical strategy can be applied generally to quantify discrete multi-component self-assembling systems.
Complex Assembly System © Copyright 2020







