
2021
23 Jul 2021, Chemical Science
Xiao Tang, Zhihao Li, Haoliang Liu, Hang Qu, Wenbin Gao, Xue Dong, Shilin Zhang, Xinchang Wang,* Andrew C.-H. Sue,* Liulin Yang, Kai Tan,* Zhongqun Tian, Xiaoyu Cao*
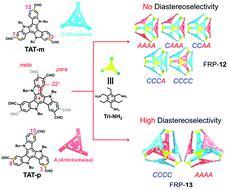
Molecular face-rotating polyhedra (FRP) exhibit complex stereochemistry, rendering it challenging to manipulate their assembly in a stereoselective manner. In our previous work, stereocontrolled FRP were gained at the cost of losing the confined inner space, which hampers their host–guest interactions and potential applications. Through a rational design approach, herein we demonstrate the successful construction of hollow FRP with high diastereoselectivity. Whereas the [4+4] imine condensation of meta-formyl substituted C3h-symmetric TAT-m and C3-symmetric Tri-NH2 led to the formation of all feasible FRP-12 diastereoisomers; the para-substituted constitutional isomer, TAT-p, exclusively assembled into a pair of homo-directional enantiomeric FRP-13-CCCC/AAAA with a cavity size larger than 600 Å3. Detailed structural characterizations and theoretical investigations revealed the thermodynamic landscape of FRP assembly can be effectively shaped by modulating the van der Waals repulsive forces among the facial building blocks. Our work provided a novel strategy towards stereospecific assembly of pure organic cages, opening up new opportunities for further applications of these chiral materials
27 Jun 2021, Giant
Liulin Yang, Pufan Ouyang, Yuan Chen, Shishuai Xiang, Yonghong Ruan, Wengui Weng,* Xumin He,* Haiping Xia,*
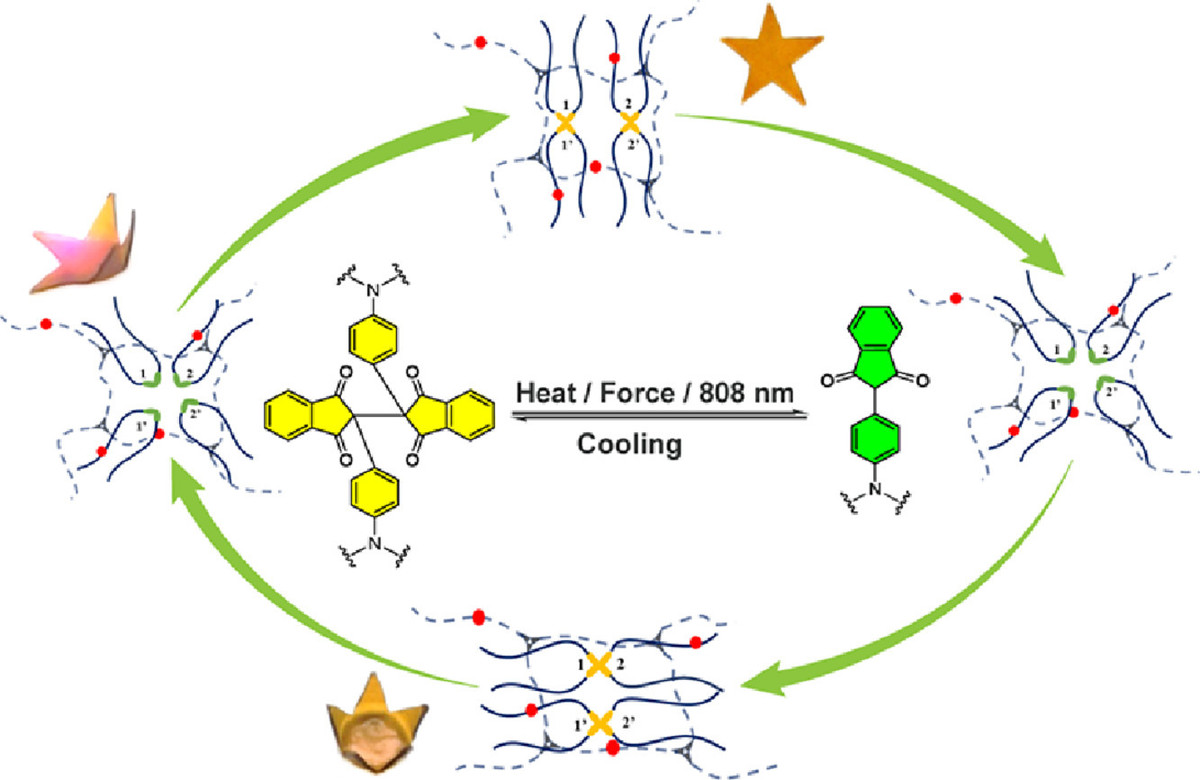
Multi-responsive polymers have attracted great attention in materials science. Herein, we report a tough self-reporting polyurethane elastomer with near infrared light induced shape memory. A radically exchangeable covalent 2,2′-bis(2-phenylindan-l,3-dione) bond is used as a dynamic covalent crosslink together with permanent crosslink and an organometallic complex carbolong 556 as a photothermal agent in the polyurethane network. The dynamic covalent 2,2′-bis (2-phenylindan-l,3-dione) bond, which is reversibly responsive to both thermal and mechanical stimuli, endows the elastomer with high toughness, mechanochromism, thermochromism, photothermochromism and shape memory behavior which can be triggered by direct heating or near infrared light irradiation mediated by the carbolong 556. Mechanochromism and thermochromism/photothermochromism are practically useful to self-reporting of mechanical failure and thermal condition, respectively. The near infrared light responsiveness facilitates a precisely spatial and temporal control of the shape memory behavior. This strategy opens up new opportunities to smart polymers.
12 Jun 2021, Advanced Materials
Longzhu Liu, Shiyan Chen, Yangyang Qu, Xiang Gao, Liang Han, Zhiwei Lin, Liulin Yang, Wei Wang, Nan Zheng, Yongye Liang, Yuanzhi Tan,* Haiping Xia,* and Feng He*
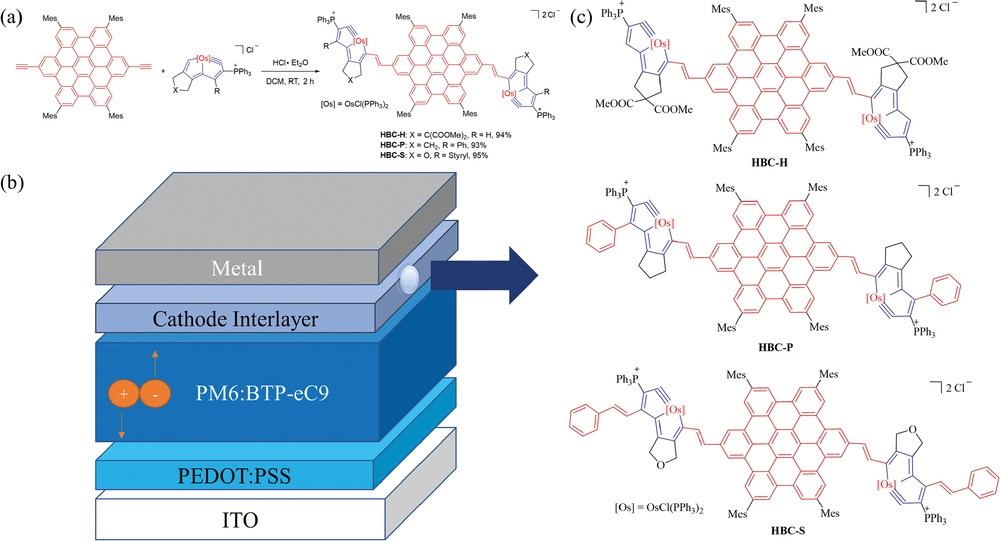
Interface engineering is a critical method by which to efficiently enhance the photovoltaic performance of nonfullerene solar cells (NFSC). Herein, a series of metal–nanographene-containing large transition metal involving dπ–pπ conjugated systems by way of the addition reactions of osmapentalynes and p-diethynyl-hexabenzocoronenes is reported. Conjugated extensions are engineered to optimize the π-conjugation of these metal–nanographene molecules, which serve as alcohol-soluble cathode interlayer (CIL) materials. Upon extension of the π-conjugation, the power conversion efficiency (PCE) of PM6:BTP-eC9-based NFSCs increases from 16% to over 18%, giving the highest recorded PCE. It is deduced by X-ray crystallographic analysis, interfacial contact methods, morphology characterization, and carrier dynamics that modification of hexabenzocoronenes-styryl can effectively improve the short-circuit current density (Jsc) and fill factor of organic solar cells (OSCs), mainly due to the strong and ordered charge transfer, more matching energy level alignments, better interfacial contacts between the active layer and the electrodes, and regulated morphology. Consequently, the carrier transport is largely facilitated, and the carrier recombination is simultaneously impeded. These new CIL materials are broadly able to enhance the photovoltaic properties of OSCs in other systems, which provides a promising potential to serve as CILs for higher-quality OSCs.
26 Apr 2021, Chemistry—A European Journal
Youzhen Zhuo, Xiuxiu Wang, Si Chen, Hang Chen, Jie Ouyang, Liulin Yang, Xinchang Wang,* Lei You,* Marcel Utz,* Zhongqun Tian, and Xiaoyu Cao*
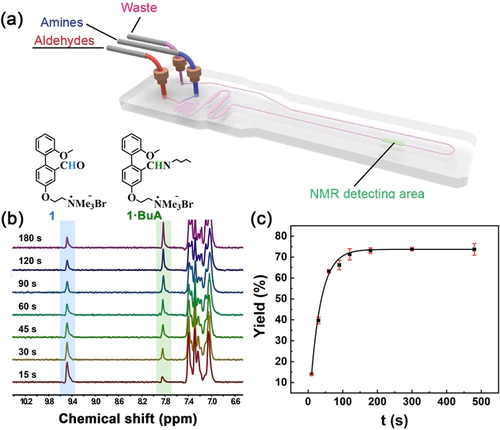
Quantitatively predicting the reactivity of dynamic covalent reaction is essential to understand and rationally design complex structures and reaction networks. Herein, the reactivity of aldehydes and amines in various rapid imine formation in aqueous solution by microfluidic NMR spectroscopy was quantified. Investigation of reaction kinetics allowed to quantify the forward rate constants k+ by an empirical equation, of which three independent parameters were introduced as reactivity parameters of aldehydes (SE, E) and amines (N). Furthermore, these reactivity parameters were successfully used to predict the unknown forward rate constants of imine formation. Finally, two competitive reaction networks were rationally designed based on the proposed reactivity parameters. Our work has demonstrated the capability of microfluidic NMR spectroscopy in quantifying the kinetics of label-free chemical reactions, especially rapid reactions that are complete in minutes.
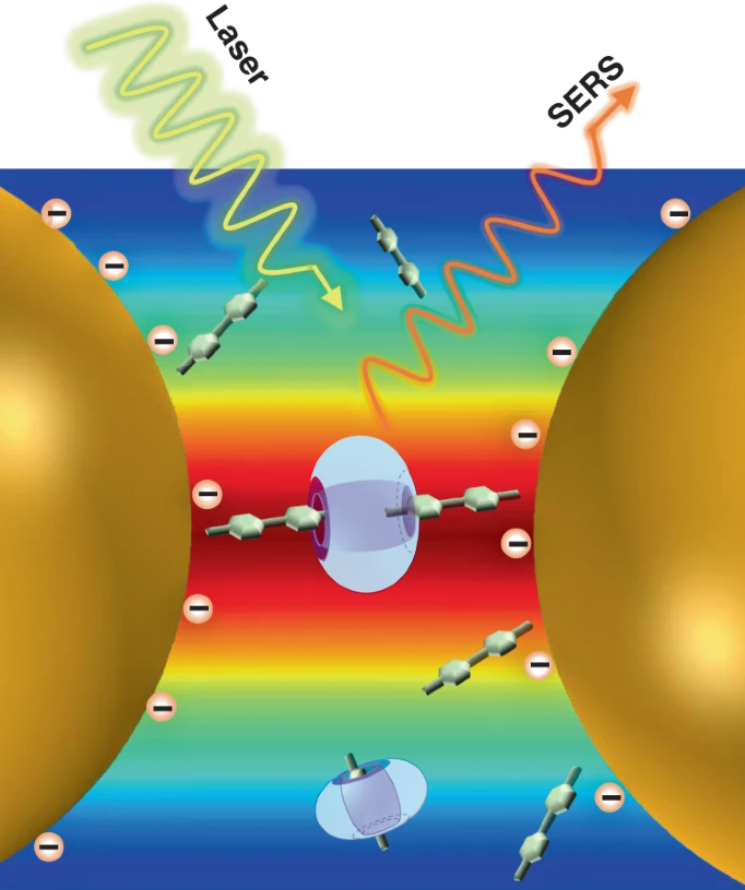
19 Apr 2021, LIGHT-SCIENCE & APPLICATIONS
Gan-Yu Chen, Yi-Bin Sun, Pei-Chen Shi, Tao Liu, Zhi-Hao Li, Si-Heng Luo, Xin-Chang Wang, Xiao-Yu Cao, Bin Ren*, Guo-Kun Liu, Liu-Lin Yang* and Zhong-Qun Tian*
Interfacial host–guest complexation offers a versatile way to functionalize nanomaterials. However, the complicated interfacial environment and trace amounts of components present at the interface make the study of interfacial complexation very difficult. Herein, taking the advantages of near-single-molecule level sensitivity and molecular fingerprint of surface-enhanced Raman spectroscopy (SERS), we reveal that a cooperative effect between cucurbit[7]uril (CB[7]) and methyl viologen (MV2+2I−) in aggregating Au NPs originates from the cooperative adsorption of halide counter anions I−, MV2+, and CB[7] on Au NPs surface. Moreover, similar SERS peak shifts in the control experiments using CB[n]s but with smaller cavity sizes suggested the occurrence of the same guest complexations among CB[5], CB[6], and CB[7] with MV2+. Hence, an unconventional exclusive complexation model is proposed between CB[7] and MV2+ on the surface of Au NPs, distinct from the well-known 1:1 inclusion complexation model in aqueous solutions. In summary, new insights into the fundamental understanding of host–guest interactions at nanostructured interfaces were obtained by SERS, which might be useful for applications related to host–guest chemistry in engineered nanomaterials.
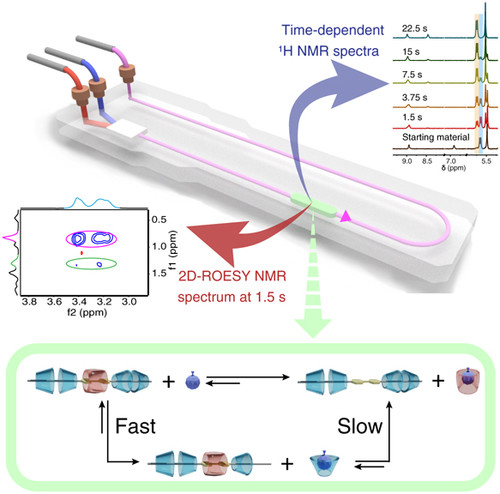
24 Feb 2021, CCS Chemistry
Yibin Sun†, Hongxun Fang†, Xujing Lin, Xiuxiu Wang, Ganyu Chen, Xinchang Wang*, Zhongqun Tian, Liulin Yang*, Marcel Utz* & Xiaoyu Cao*
It is challenging to investigate fast supramolecular processes due to the lack of appropriate characterization methods with high structural resolution. In this study, microfluidic nuclear magnetic resonance (μF-NMR) spectroscopy was employed to monitor the kinetics of threading and dethreading of a pseudo[6]rotaxane system. By employing high time resolution μF-NMR, 1H and two-dimensional (2D) rotating frame nuclear Overhauser effect spectroscopy (ROESY) NMR spectra were recorded at any time point within 1.5 s after the onset of the process. This technique enabled the successful identification of kinetic intermediates and rate-determining steps, usually impossible to determine by other spectroscopic techniques. Thus, our study demonstrates the capability of μF-NMR in investigating the mechanism of complex supramolecular systems.
Complex Assembly System © Copyright 2020





